
Placing products on the EU and or the UK market requires a local entity that can take care of questions asked by market surveillance authorities.
If a product is not marked with the address information of a local entity, authorities will decide who is responsible for product compliance. For example, it could be your customer, the shop selling the product, or the fulfilment service provider you use to sell products directly to consumers.
Most likely, neither would like to be placed in that role. Especially fulfilment service providers do not want to become responsible for product compliance and push sellers to find a solution. Setting up your local entity is a solution. However, it takes time and can prove to be costly. Note that a simple “PO Box” firm is not enough!
Luckily there is also an efficient alternative; assign ProductIP to act as your European and or UK Authorised Representative.
Your distributors and agents can then focus on sales of products carrying your brand. ProductIP focuses on you, your products and requirements. ProductIP takes care of your interests. We offer stress-free product compliance.
European CE Marking Directives, and UK UKCA Marking Regulations, place the responsibility for compliance on the manufacturer of the product concerned. Even where the manufacturer is located outside the European Union or the UK, and therefore out of legal reaches of the EU or UK enforcement authorities, the manufacturer has certain obligations (e.g., quality control), which they cannot assign to other parties. The EU considers products underway to consumers as “being made available” and therefore they need to comply with all the requirements.
Moreover, in cases where the manufacturer is not based in the EU or the UK, legal responsibility for compliance with the directives lies with the entity responsible for selling the goods within the EU or UK, unless the manufacturer has appointed an Authorised Representative. The manufacturer can then assume his responsibility and give a mandate to ProductIP as an Authorised Representative to represent the manufacturer towards authorities.
The need for, and obligations of, Authorised Representatives are much misunderstood. All CE marking directives (in EU), and UKCA Marking Regulations (in UK) place responsibility for compliance on the manufacturer of the product concerned. These are the three options:
Option 3 is the ideal option for Sellers shipping to the end user via online platforms, regular import and trading channels or directly to consumers.
Note: The ProductIP Authorized Representative service is also available for brand owners already located within the EU / EFTA or UK. The benefits regarding info on labels, etc. are identical.
The local entity requirement applies also for EU companies selling in the UK, and for UK companies selling in the EU. Northern Ireland continues to follow the European Framework.
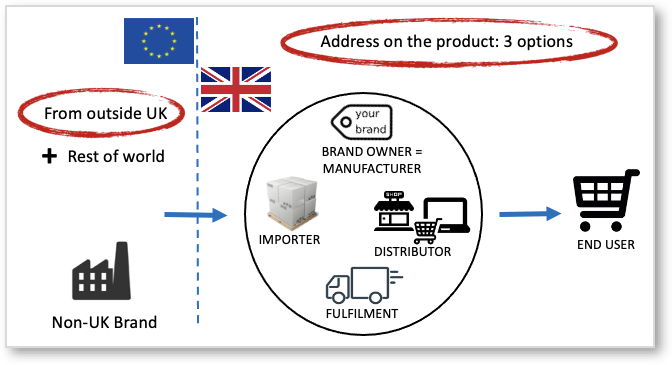
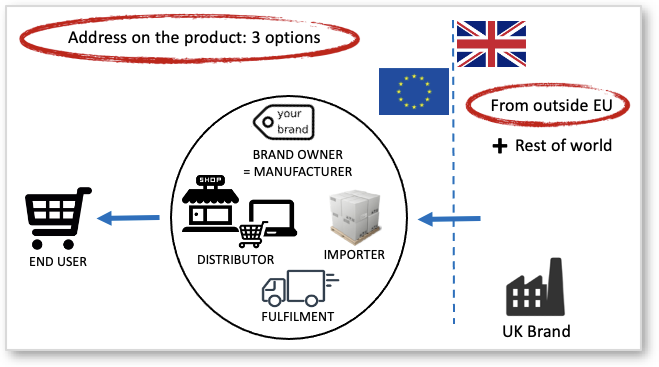
If the manufacturer is not based in the EU / EFTA, or not based in UK then legislation allows that their ‘Authorized Representative’ takes care of certain duties related to product compliance. Some call it “Responsible Person” but that is another obligation connected to specific product groups such as cosmetics and medical devices. ProductIP is not a Responsible Person as such. Note that this term is often misused in the market causing a lot of confusion.
The scope of the regulation EU 2019/1020 is limited to harmonised goods, resulting in application of CE marking. However, Germany extended the scope when transferring it into national law (ProdSG). In Germany it now also covers non-harmonised goods. Given the fact that the German market plays a significant role in the world of e-commerce this will have impact on the total market.
For the UK market it is relevant for all non-food products or as the UK legislation calls it: manufactured goods, including products that do not need to carry a CE marking.
The ProductIP Authorised Representative Service includes:
Additionally, ProductIP may arrange (through a third party) for you to deal with: *
* Additional charges will be applicable
ProductIP holds no liability for any damages that occur from placing the products on the market. ProductIP holds no liability for any damages that occur from claims regarding patents and technology licenses.
Besides the annual fee to maintain your authorized representation we will provide you with a specified monthly financial statement for hours and expenses that occurred. Next to the annual fee, we charge you a one-time initial payment for 8 hours. We will ask upfront approval for events for which we expect the hours involved to exceed the initial payment. Mandatory usage of the ProductIP platform and creation of files are not included in this fee.
What do we expect from you as a responsible Manufacturer/Seller?
We need you to
For this purpose we conclude with you an EU or UK Authorised Representative Service Agreement. It just takes three steps.
Click here to download the agreement text for EU Authorized Representative
Click here to download the agreement text for UK Authorized Representative
STEP 1. Create your account in ProductIP, you can digitally sign the Authorized Representative Agreement. It is a clear step-by-step process.
STEP 2. We will raise an invoice and e-mail that as PDF. Settle the related annual fee to activate the agreement.
* You will be invoiced in Euro’s from the Netherlands. Or in HK$ or US$ from Hong Kong.
STEP 3. Buy credits so you can start making technical files in ProductIP. Adapt your artwork with the new address information and support compliance via files in your ProductIP account*
* Technical files need to be created in ProductIP. For this service several support and pricing models exist. click here for the ProductIP Service Overview
Welcome to stress-free product compliance. Continue increasing your business.
The ProductIP Authorised Representative services are possible for non-food products (manufactured goods) that are under the scope of one or more of the following EU directives/regulations and their UK equivalents
| EU | UK |
| General Product Safety Directive (General Merchandise) | General Product Safety Regulations (General Merchandise) |
| Framework for Food Contact Materials | The Materials and Articles in Contact with Food Regulations |
| Low Voltage Directive | The Electrical Equipment (Safety) Regulations |
| Electromagnetic Compatibility Directive | Electromagnetic Compatibility Regulations |
| Personal Protective Equipment Directive | Personal Protective Equipment Regulations |
| Toys Directive | Toys Regulations |
| RED Directive | Radio Equipment Regulations |
| Machinery Directive | Supply of Machinery Regulations |
| Construction Products Regulation | Construction Products Regulations |
| RoHS (not including WEEE services!) | RoHS Regulations |
| Cosmetics Regulation (Not Responsible Person services!) | Cosmetics Regulations |
| Detergents Regulation | Detergents Regulations |
| Pressure Equipment Directive | Pressure Equipment Regulations |
| Outdoor Noise Directive | Outdoor Noise Regulations |
| Automotive regulations | The Road Vehicles Approval Regulations |
| Measuring Instruments Directive | Measuring Instruments Regulations |
| Non-automatic Weighing Instruments Directive | Non-automatic Weighing Instruments Directive |
| Aerosol Dispensers Directive | Aerosol Dispensers Regulations |
| Medical Device Regulation (ONLY class I, non-sterile) | Medical Devices Regulations (ONLY class I, non-sterile) |
And various other applicable regulations as generated from the ProductIP regulatory database. Involvement of Notified Bodies (EU) or Assessment Bodies (UK) may be required in order to comply with the essential requirements. ProductIP does not provide Responsible Person services. ProductIP does not provide a post-market surveillance system. Excluded are medicines, products that are also covered by the Medical (Sterile, Class 2 and 3) and/or Pyrotechnic Products and/or ATEX Directives and/or Annex IV of the Machinery Directive and living organisms, and their UK equivalents.